Tags
aquaculture, cytochalasin B, huître des quatre saisons, meiosis, polar body, polyploidy, tetraploid oysters, triploid oysters
Long ago were the halcyon days of seas, brimming with all kinds of fish and molluscs, and laissez-faire politics that shunned any form of regulation on fishing, best exemplified by the opinions of one of the intellectual giants of the 19th century, Thomas Huxley, an autodidactic natural scientist, who dubbed himself “Darwin’s bulldog” because of his staunch defence of the latter’s controversial ideas, and who fathered a long line of Huxley geniuses. In his opinion, the bounties of the oceans were considered inexhaustible and nature left to its own devices, in the true spirit of free trade and liberalism, was almost infinitely resilient and could adapt to any pressure imposed by man so that the idea of any threat of overfishing was totally rejected. To be fair to Huxley, towards the end of his life, his tone changed, as he became more convinced that management of oyster beds needed to be introduced and recognised the dangers inherent in certain practices. Apart from France, most countries failed to introduce any viable system of regulation until it was too late. Nowadays, the tragedy of all this naivety, on the one hand and greed, on the other, is only too apparent. Stocks have fallen dramatically and fishing has now become far more regulated. Aquaculture is seen as an economic and environmental necessity to safeguard the finite resources of the seas. However, not all aquaculture is sustainable, and in a recent book by Colin Nash, The History of Aquaculture (2011), a pile of evidence is amassed of the unsavoury involvement of the nuclear power industry and multinational chemical conglomerates like Union Carbide, Dow Chemical and Sun Oil in aquaculture during the 1960/70’s which had devastating consequences for the marine environment. Aquaculture was seen as a way to buy good publicity and acquire a brand as a caring company.
Right from the beginning, science has endeavoured to involve itself in aquaculture. One of its pioneers, known in France as le père de la pisciculture, Victor Coste (1807-1873) was originally professor of embryology, and was instrumental in spreading interest in the methods of artificial collection of wild spat from oysters. His was the age of the first hatcheries which were established to study and allow fish spawn in artificial environments. But science was generally slow to latch on. The first experimental hatcheries on a larger scale were started in the 1930’s, in Conwy, Wales (UK) under first Herbert Cole (1911-1984) and later Peter Walne (1926-1978) and in Milford, Connecticut (US) under Victor Loosanoff (1899-1987). But most of the work only got off the ground after the 2nd World War. Selective breeding and artificial rearing of oyster spat in hatcheries were seen as ways to compensate for the disappearance of wild stocks and unpredictability of spawning in colder climates by providing an almost limitless source of spat for cultivation. The first commercial oyster seed hatchery opened on the US west coast in 1967, but like most hatcheries was beset with various biological problems.
So this was the scene when a young and ambitious student set about trying to create a hybrid oyster, one which had never existed in nature. The narrative begins in a wooded, hilltop research centre, now the Ira C. Darling Marine Center, overlooking the Damariscotta River, on the Maine coast in North Eastern America, where in 1979 marine biologists at the University of Maine were working on methods to help improve the local shellfish industry. It was important to find ways to make fish grow more quickly in the colder waters, to overcome the problems of erratic spawning at such low temperatures and to make more money by producing shellfish for consumption the whole year round. The idea of growing brood stock in hatcheries was not new but producing a sterile oyster was, one that would be denied nature’s most basic function, reproduction, so that meat content, flavour and texture could be improved. Instead of utilising its sugar reserves of glucose and glycogen for gamete production, and reducing its meat content by as much as 70%, the sterile oyster, it was thought, could be freed to harness this energy for meat and shell growth, thus reducing the time to cultivate a marketable oyster. Another benefit in a faster growing oyster was that it could reach market size before being vulnerable to particular types of parasites like the one causing Dermo disease (Perkinsus marinus). In a word, the sterile triploid was going to be created because it made irrefutably marketing sense.
The Ira C. Darling Marine Center at the University of Maine
The story of the triploid oyster is a fascinating and to some extent frightening chapter in the history of aquaculture. It epitomises man’s desire to master and rise above the unpredictability of nature, but it also poses uncomfortable questions about the lengths to which man has gone in the pursuit to modify the ecology of nature. As Sir Maurice Yonge (1899-1986), a distinguished marine zoologist of his day, wrote in his Oysters on the future of oyster culture, “the more man interferes with nature the greater become the problems he creates (1960, 189).
Some elementary facts about genetic biology may be needed here. In the animal kingdom, nearly all species are diploid, that is, each of their somatic cells contains two sets of homologous chromosomes, one each from the male and female parent. Somatic cells give rise to the development of the individual body through the process of mitosis, in which cells divide through DNA replication, thus retaining their two sets of identical chromosomes. Germ cells, which are responsible for reproduction and formation of a new generation, are located in the gonads and develop into male and female gametes (i.e. sperm and egg). This process whereby germ cells recombine their genetic DNA molecules of homologous chromosomes (synapsis) and lose one of their sets of chromosomes and become haploid (a single set of chromosomes) progeny cells or gametes is called meiosis. As a couple of genetic biologists[1] so succinctly wrote, “the very essence of sex is meiotic recombination.” (We never learnt that at school!). Meiosis involves intricate phases of chromosomal separation, rearrangements and segregation before new haploid cells are formed, all within a relatively short period of time, although it is divided into two main stages, meiosis I and II. In each of these two stages, crucially so-called polar bodies are extruded (released) and serve as biological indicators of the development of meiosis, especially in the creation of triploid egg cells. However, the process of meiosis in many marine molluscs, including oysters is delayed and only completed after fertilisation, whereas in most other animals this process is achieved before fertilisation. It is this complicated and amazing process of meiosis that is manipulated, by inhibiting or blocking the release of the polar bodies either in meiosis I or meiosis II, in order to ensure that the egg retains its two sets of chromosomes. Normally, one set of chromosomes would be shed to make way for the set of chromosomes provided by the male sperm to secure the continuation of diploidy in the organism. If this manipulation succeeds, then the fertilised egg contains three sets of chromosomes, that is becomes a triploid cell, which then can undergo mitosis in the usual way. It was generally assumed that adult triploids were sterile since their three sets of homologous chromosomes could not successfully recombine during meiosis.
In humans and mammals generally, the condition of triploidy is always life-threatening, if not lethal, but in the non-vertebrate and plant world, there are many species, which exist in natural states of polyploidy (several sets of chromosomes). For instance, there are wild species of berries belonging to the genus of Vaccinium, like blueberries, cranberries and lingonberries that are polyploid (tetraploid and hexaploid), as well as diploid. There are even varieties of grapes that have been discovered to have this feature. Some common agricultural fruits, such as melons, bananas and oranges have also been manipulated into polyploids to grow bigger and more quickly.
Meanwhile back in Maine, research was geared to creating polyploid shellfish, and after a series of trial and error experiments, one technique, which had been used on clams as well as salmon and rainbow trout in Norway earlier in the early 1970’s, was selected with its fair share of serendipity. It involved the insertion of a toxic chemical, a mycotoxin, cytochalasin B, at a critical moment during meiosis into the newly fertilised egg to prevent the reduction of the two sets of the female chromosomes to one, so that it would end up with three sets (triploidy). Timing, duration and dosage levels were crucial and could in worst cases cause genetic abnormalities (aneuploidy) and high mortalities at various stages of larval development. The optimal point when the toxic chemical was inserted was during meiosis II, to inhibit the release of the second polar body and thus produce a triploid zygote (fertilised egg).

The development of chemically induced triploid zygotes during meiosis II
This laboratory technique of using cytochalasin B was gradually perfected and ushered in a new era in oyster cultivation, in which an artificial, supposedly sterile species, not genetically modified however, the triploid, could be used to produce a more meaty and juicy oyster more quickly, and even during the summer, “r-less” months. The young graduate student behind this work was Standish K. Allen Jnr, who together with his supervisor Herb Hidu and mentor Jon Stanley, is credited with the innovative research, conducted with the Eastern or Atlantic oyster, Crassostrea virginica, although he did not bother to get his “invention” patented. Their paper[2] in 1981 already mooted the idea of creating oysters with an even number of chromosome sets, like tetraploids (four sets), which then could synapse and be fertile. However, the local oyster farmers in Maine were too conservative then to embrace this new technology and the hatcheries that existed were small and more experimental than commercial.
So Allen jetted off in 1983 instead to the Northwest, eventually to complete his doctoral studies with a well-known biologist in the field, Kenneth Chew, in Seattle, where the oyster industry was far more commercialised, and ready at work on the Pacific oyster, Crassostrea gigas. Since this latter oyster generally was unable to spawn naturally in the colder Pacific water, well-established hatcheries had already begun to produce diploid oyster seed for cultivators to grow. He and another researcher, Sandra Downing, successfully applied the technique in 1985 to large batches of oysters in a commercial hatchery setting, whose owners wanted the process patented. The patent was in due course refused on the grounds that an earlier publication (in 1981) of the process meant that it was no longer original. The end result of the application in 1987, however, did create a historical precedent, as a landmark court case, since it was admitted for the first time ever that patents could be granted to new species of animals, genetically altered or modified by science. Suddenly, the door to the world of modern biotechnology was thrown open wide by this ruling.
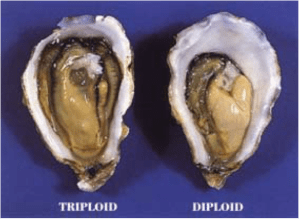
Even so, health concerns about the carcinogen, cytochalasin B, were growing, because of its links with cancer and the FDA (the Food and Drug Administration) was debating whether to ban its use in commercial hatcheries. The two researchers decided to try another method to produce triploids by subjecting oyster eggs to hydrostatic pressure, and this time their patent application was accepted. Another method that was also used was subjecting the onsetting phase of meiosis to temperature extremes. An alternative to cytochalasin B has been the use of an enzyme inhibitor, 6-dimethylaminopurine (6-DMAP). However, the downsides of these four forms of induced triploidy was that they resulted in high mortalities of the oyster larvae in the hatcheries due to the severity of the treatment, that the success rate varied and that some triploid oysters were unstable enough to revert back into diploids as they grew or were able to spawn themselves, and so were not wholly sterile. There were other contradictions that triploids produced earlier in meiosis (so-called meiosis I) grew faster but were liable to higher mortalities than triploids produced later during meiosis II. But faster growth could also have been due to the fact that triploid cells were 33% larger in volume than diploid cells. Since the whole process was fraught with risks and problems, other ways were sought.
Differences in growth between a triploid and diploid oyster after 36 months
Help came from another non-native source, a Chinese geneticist, who emigrated to Seattle in 1985 to pursue postgraduate work, Ximing Guo, and he wanted to go a step further and create a tetraploid oyster (with four sets of chromosomes) which if breeded with a natural diploid would then produce a “natural” triploid, thus avoiding the use of any toxic and cancerous chemical. The problem was that the diploid egg normally was too small to hold two extra sets of chromosomes and all his attempts ended in failure. Meanwhile, Standish Allen had relocalised back to the East coast and gained his first full-time academic post at Rutgers University and its Haskins Shellfish Research Laboratory in 1989. Within a few years, he managed to persuade Guo to join him there and the two started working together on the specific problem of creating a fertile triploid with large enough eggs, although from the outset triploid oysters were supposed to be completely sterile and unable to develop gametes. However, it was occasionally observed that such fertile triploids did exist. So once these triploid oysters and their large eggs were identified, Guo and Allen still resorted to cytochalasin B to ensure that the triploid eggs could be manipulated during meiosis I to accommodate another set of chromosomes from male diploids and then grow into oyster spat. It was found that it was absolutely necessary to monitor the timing of biological indicators in the actual meiotic events in the individual triploid female eggs rather than to follow more general criteria, if tetraploids were to be bred successfully, because of greater variability and asynchrony of triploid eggs than in diploid equivalents. Even then the average success rate after eight days was about 12% (though others have reported much lower figures), and the vast majority of the fertilised eggs were deformed aneuploids. Other critical parameters were salinity and temperature levels and the length of time spent by the eggs immersed in seawater. According to one paper written by these two scientists and two Chinese colleagues[3], the major cause for the formation of tetraploids was a mechanism during a crucial stage of meiosis II, called united bipolar segregation, when the homologous chromosomes are segregated into different cells. It is quite an ironic quirk of nature that the supply of sterile oysters depends on those very same oysters not being sterile at all!

The production of natural triploid zygotes using tetraploid males and diploid females
In 1993, the new tetraploid oyster was created in the laboratory by Guo and Allen: this was the second time Allen had invented an artificial oyster, but now he wasn’t going to miss out on creating a patent for his work. When the supply of tetraploid oysters could be regularly guaranteed, they could be used, more often than not the male species, on a large scale to breed with female diploids so as produce “natural” triploid offsprings to be used for cultivation. These “natural” triploids were after only 9 months of growth as much as 50% larger than normal diploid oysters, which satisfied both the scientists and cultivators alike, and even a third larger than induced triploids. Because of the growing dependency of the oyster industry on hatcheries for supplying oyster seed of Pacific oysters, Crassostrea gigas, there has been a rapid response from both growers and hatcheries to develop the techniques of tri- and tetraploidy, especially the West Coast of North America. Now most of the oyster seed supplied by commercial hatcheries for cultivation there are triploids, produced with the various methods described, although batches produced with older methods often may contain diploid oysters.
| United States Patent |
5,824,841 |
| Guo , et al. |
October 20, 1998 |
Tetraploid shellfish
Abstract
Provided by this invention are novel tetraploid mollusks, including oysters, scallops, clams, mussels and abalone. Also, provided are a method for producing the tetraploid mollusks and a method for producing triploid mollusks by mating the novel tetraploid mollusks with diploid mollusks.
| Inventors: | Guo; Ximing (Glassboro, NJ), Allen, Jr.; Standish K. (Mauricetown, NJ) |
| Assignee: | Rutgers, The State University of New Jersey (New Brunswick, NJ) |
| Appl. No.: | 08/895,077 |
| Filed: | July 16, 1997 |
The patent (United States Patent 5824841) was accordingly granted in 1998 to both Guo and Allen. They went on to set up a special start-up company for the creation of tetraploid molluscs with Rutgers University, 4Cs Breeding Technologies, Inc, which supplies its patented tetraploid oysters to licensed hatcheries wanting to breed 100% guaranteed triploids for cultivation.
So now this is the most common way of producing oyster triploid seed in hatcheries for the oyster cultivation, and this dependency on tetraploid technology has been growing by the year, especially in North America. Allen has continued to work on producing disease-resistant strains of tetraploids and it is easy to see how the research conducted by him and others, for instance, now at the Aquaculture Genetics and Breeding Technology Center within the Virginia Institute of Marine Sciences, on chromosome set manipulation will eventually lead, if not already, to genetic selection, to the development of specific strains of triploid oysters which not only grow faster and bigger, but will also have particular shell characteristics and be able to resist viruses, parasites and pollutants and no doubt even in due course – to the area of transgenics and genetic modification where DNA material from another species is introduced. In addition, there are concerns about the long-term risks over generations of using a mycotoxin, like cytochalasin B, in the creation of first-generation tetraploids, as very little is known about such effects.
Oysters have always been considered, like many other shellfish, as one of the last natural products and have often been marketed as such. If they gradually lose not only this status and also reputation, there may be consequences for their consumption. Fortunately there are stocks of wild oysters still being cultivated and even seed from these stocks, which is sold to other growers and hopefully this will continue and be preserved.
France is another country which has taken on board the benefits of growing triploids, known there as l’huître des quatre saisons – the oyster for the four seasons. Ever since 1997 when IFREMER – a State research institute for marine exploitation – purchased tetraploid oysters to breed, many cultivators have been enthusiastic about buying oyster seed from its hatcheries, which became commercially available 2000. However, ethical controversies still arise about their place and effects in the biological diversity of marine ecosystems and also among consumers who are sceptical to the product.
However, on the other hand, science and man are doing all they can to eclipse nature, but nature will have the last say or laugh whatever and man will always be playing a desperate catch-up game in which the rules are surreptitiously altered and which will probably lead us into an irreversible cataclysm. Already it is estimated that 85% of all native oyster reefs have been made extinct globally, and in many areas the loss is more than 99%[4]. But it is not just the reefs that have disappeared but probably more importantly entire marine ecosystems that the oysters basically provide: such services as water filtration, food and habitat for other species and coastal stabilisation and defence. If sustainability in oyster fishing is to be achieved, reef conservation and management need to be strictly enforced, including the establishment of protected areas and the ban of destructive harvesting practices. A concerted and joint effort from various stakeholders, such as fishermen, aquaculture companies, public agencies, environmental and conservation groups and other NGOs, is absolutely necessary if a long-term rebuilding of oyster reefs and sustainable harvests is to be achieved, rather than the short-sighted goals of put-and-take fishing that has often happened. But all this goes against the grain of the ways and shifts of a life of autonomy that have marked fishermen, watermen and sea-faring communities for centuries; they now also have had to resist being overwhelmed by urbanisation, gentrification and industrialisation. And they have seen the source of their livelihood invaded and taken over by conglomerates and with their backs against the wall have become all the possessive about their marine territory, possibly as a last desperate measure to safeguard its dwindling riches. In a way, who can blame them? Rather, it has been the inevitable spread of urbanised life in all its avatars that has killed the oyster beds, the frenzied demand and over-consumption, disease, pollution and acidification – in a couple of words, modern civilisation. So it is now down to those most exemplary carriers of the latter, the scientists, to come up with laboratory solutions that will repair and restore the depleted oyster banks that once filled our coastal waters.
[1] Villeneuve, A.M. & K. J. Hillers: Whence Meiosis? Cell, 106 (2001), 647-650.
[2] Stanley, J.G., S. K. Allen and H. Hidu: Polyploidy induced in the American Oyster, Crassostrea Virginica, with Cytochalasin B. Aquaculture, 23 (1981), 1-10.
[3] Que, H. et al: Chromosome segregation in fertilised eggs from triploid Pacific oysters. Crassostrea gigas (Thunberg), following inhibition of polar body 1. Biological Bulletin, 193 (1997), 14-19.
[4] Beck, M.W. et al:_Oyster Reefs at risk and recommendations for conservation, restoration and management. Bioscience, 61 (2011), 107-116.